|
Introduction to the Definitive Guide to ISO 14971 Risk Management for Medical Devices. My entry into the medical device industry was not a planned career path. Within the first few months of starting as a product development engineer, I knew that I would spend the rest of my life involved with the medical device industry.
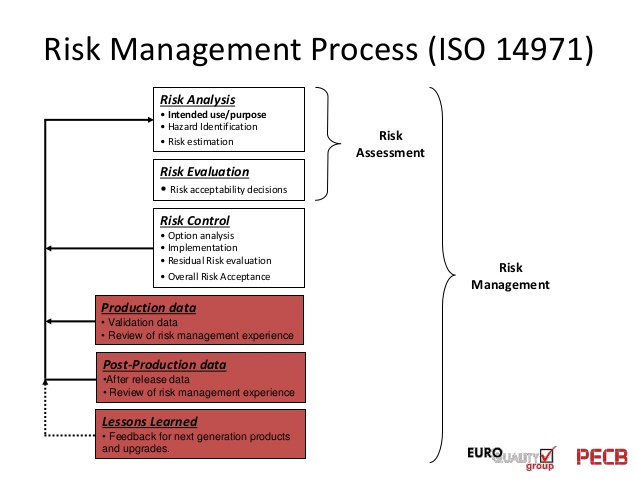 General information
Life cycle
Skype ipad download. A standard is reviewed every 5 years
Risk Management Standard Iso 14971Revisions / Corrigenda
+41 22 749 08 88
Iso 14971 Ppt
Opening hours:
Monday to Friday - 09:00-12:00, 14:00-17:00 (UTC+1) Keep up to date with ISORisk Management Standard Iso 14971
Sign up to our newsletter for the latest news, views and product information
Comments are closed.
|
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Blog
- Desert Storm Game Download Free
- Vietnamese Font For Windows 10
- Games App Download For Pc
- Activate Windows Watermark Remover
- Htc M9 Software Update
- Galaxy Tab A Usb Driver
- Wizard101 Free Membership Code
- Boot Manager Download Free
- Simcity 4 No Cd Crack
- Best Hidden Object Game Sites
- Bangla Kobita Free Download
- Alphablocks Game Bbc Uk
- Leappad Ultimate Free Code
- Free Torah Bible Code Software
- Java Bangla Book Download
- Skype Ipad Download
- Dungeon Keeper Gold Free Download
- Singer Serial Numbers And Models
- Update My Tomtom For Free
- Download Microsoft Office 2007 Crack Version
- Adobe Indesign Templates
- Pinnacle Dazzle Software Free
- Adobe Premiere Pro Cc Crack
- Windows Compressed File Password
- Foxpro Download Free
- Radeon Xpress 200 Windows 10
- Linux Security Dvr Software
- Tales Of Vesperia Gameplay
- Windows 95 Image File
- Xpadder Windows 10 Xbox
- Install Winusb Driver
- Game Pc Computers
- Win 7 Download Torrent
 RSS Feed
RSS Feed
